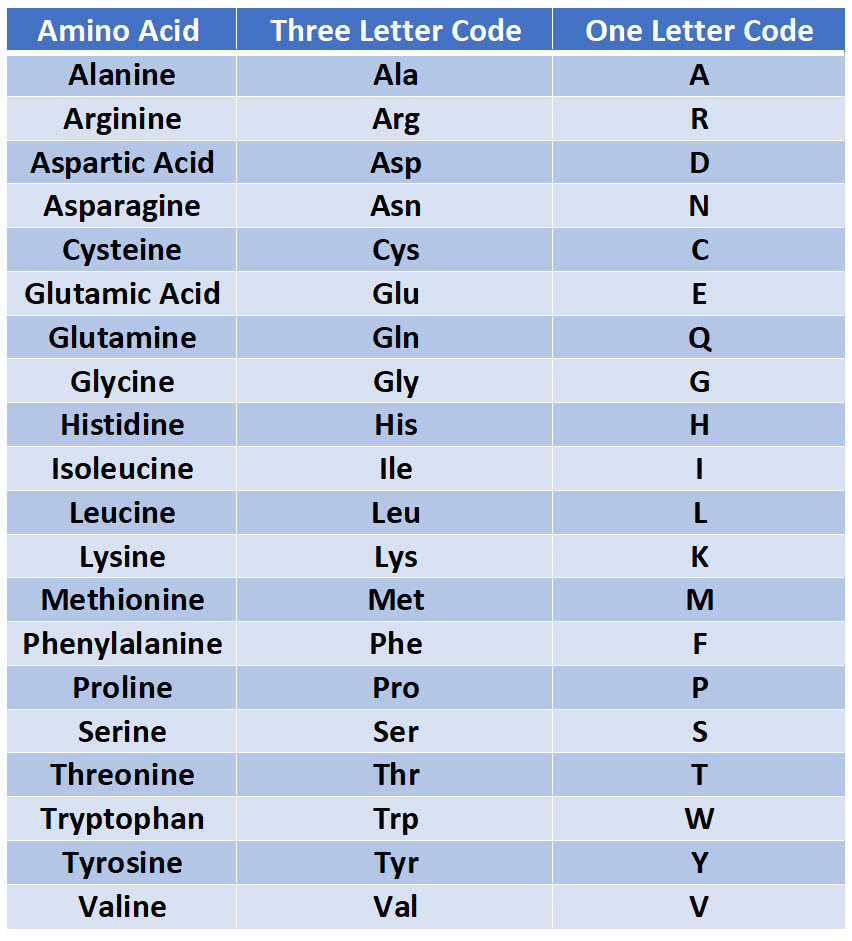
The so-called salt bridges, which are formed by the interaction between positively and negatively charged amino acid side chains, have been found to be important for the stabilization of protein three-dimensional structure. There are four of them, two basic amino acids, lysine (Lys) and arginine (Arg) with a positive charge at neutral pH, and two acidic, aspartate (Asp) and glutamate (Glu) carrying a negative charge at neutral pH. 
It is easy to see which amino acids are charged simply because at neutral pH (around 7) they contain a single charge. For example, based on the propensity of the side chain to be in contact with water, amino acids can be classified as hydrophobic (low propensity to be in contact with water), polar and charged (energetically favorable contacts with water). Each of the 20 most common amino acids has its specific chemical characteristics and its unique role in protein structure and function.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
